 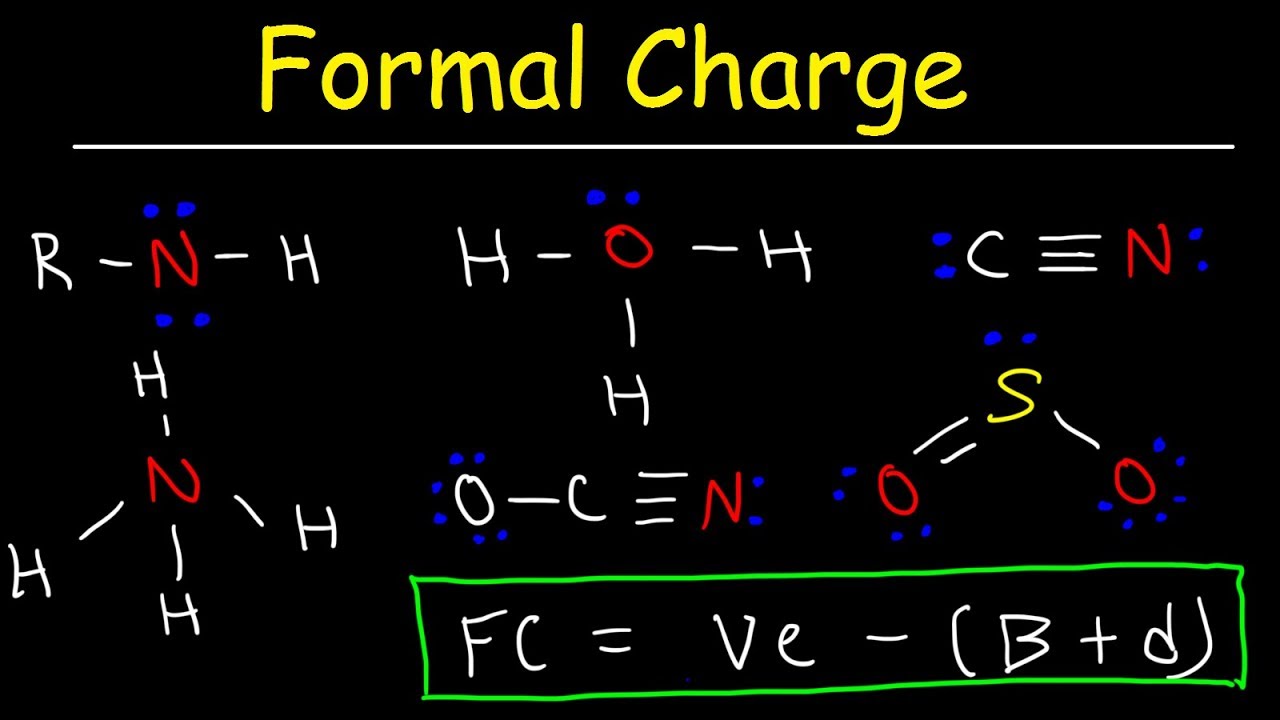 
Want to see this shortcut brought to life? See my formal charge video below Lone pairs represent 2 electrons sitting on the atom so that Has = 2Įach bond only counts for a single electron since the second electron in the bond is touching the other atom. Has = the number of electrons an atom HAS directly attached, touching the atom in question. Should = the number of valence electrons that a neutral atom SHOULD have. Let’s make sure you understand this shortcut This shortcut is guaranteed to save precious seconds on your exam IF AND ONLY IF you understand how to apply it.īut when you understand it you’ll be able to solve formal charge in your head, in under 8 seconds per atom. Here another option, MY version – one that is easier, faster, and comes out with the same result! Formal Charge Calculation Shortcut Yes the equation works, but it’s far too tedious and annoying! There are just too many steps and calculations. A triple bond of 6 electrons allows each atom to claim three.ĭo you dread running through this every time you finish a reaction? A double bond containing four electrons allows each atom to claim 2. ½ # bonding electrons = Since a bond is formed by sharing 2 electrons between 2 atoms, every atom in the bond can only take credit for one of the 2 electrons.Each electron counts as one and so a pair counts as two. Lone Pairs = lone electrons sitting on the atom.Valence electrons = corresponds to the group number of the periodic table (for representative elements).Let’s start with the proper formula and then learn a shortcut. A long, annoying trip to my professor’s office hours enabled me to figure it out, but it was still overwhelmingly TEDIOUS! The formula in my textbook was long, tedious and brutal. When I first studied formal charge I was lost. Now that you know WHAT we’re studying, let’s see HOW! – That’s where I can help you. 
If the atom loses negative electrons and therefore has as few as one extra POSITIVE protons in its nucleus, it will carry a net positive charge. If the atom has just one more negative electron, than protons, it will have a net negative charge. An ion is simply an atom or molecule that gained or lost electrons to get a net charge. Think back to general chemistry when you studied ion formation. The answers are yes, yes, yes, and more yes. Which specific atom is responsible for the overall charge?Īnd, what if you’re studying an uncharged molecule – Is it still possible for individual atoms to carry charge despite the net neutrality? Even the negative charge on the hydroxide oxygen is simple to understand.īut what if you have a much larger group of bound atoms with an overall net charge? For example, the negative nitrate or triple negative phosphate. Chloride obviously has a negative charge. This concept is simple enough for small ions. The sum of formal charges on any molecule or ion results in the net overall charge. 
So what exactly IS formal charge?įormal charge is the actual charge on an individual atom within a larger molecule or polyatomic ion. Formal charge helps you understand reaction patterns by showing you why a specific atom attacks, and why its ‘victim’ accepts the attack. Yet understanding the nature of Formal Charge is a critical component when it comes to mastering organic chemistry reactions and mechanisms. How can you afford that kind of time when working through a multi-step synthesis? Formal Charge is that pesky concept covered early in organic chemistry and somehow never seems to go away.īecause the equation in your textbook is long, confusing, and needlessly annoying. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
